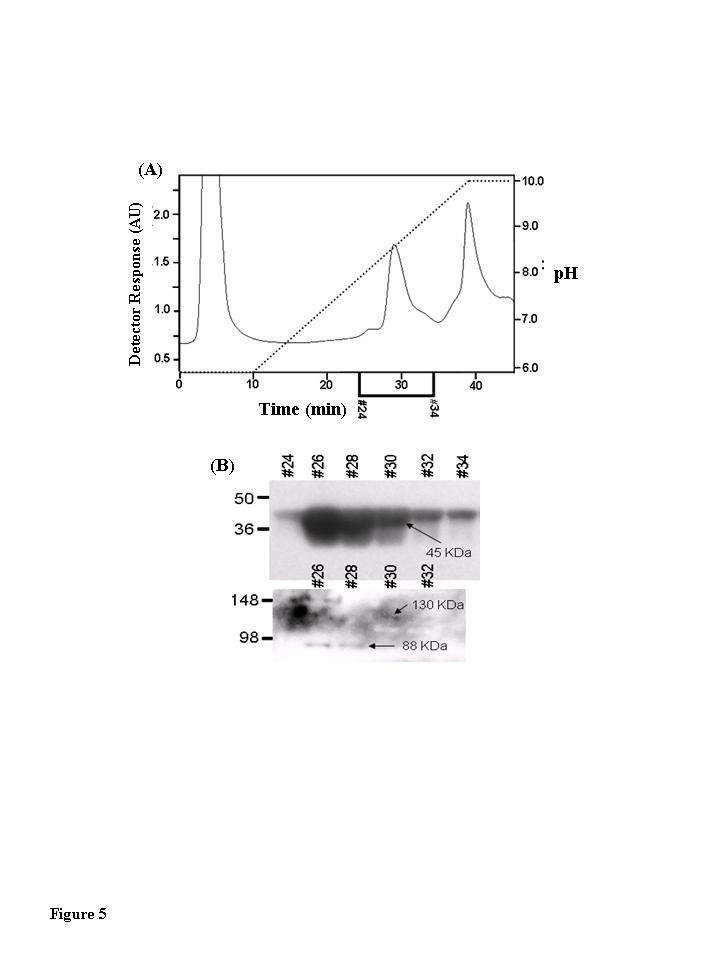
Figure 5.Muscle creatine kinase exists as 130 kDa and 88 kDa species in vivo. Blue Sepharose
affinity column fractions of cytosolic protein from quadriceps of young,
middle-aged, and aged mice that contained CKm were identified by Western
blotting. The samples were further fractionated by hydroxyapatite (HA)
chromatography. (A) HA fractionation of Blue Sepharose cytosolic
quadriceps fractions from Blue Sepharose fractionation. After application
of samples to the HA columns, the loaded columns were washed with 10 ml of
50 mM sodium phosphate pH 5.8 and developed with a 30 ml linear pH
gradient (pH 5.8 to pH 10.0). Flow rate equaled 1 ml/min throughout
purification and fractions were collected at a rate of one fraction per
minute. The chromatograph in this figure represents fractionation of the
middle-aged protein sample. Fractionation of the young and aged samples
yielded similar chromatographs. (B) Even numbered HA fractions
(26-34, 20 μl) were resolved
under denaturing conditions by SDS PAGE and transferred to a PVDF membrane.
Blots were probed with an antibody specific for CKm. A short exposure (15
second expo-sure, top blot) reveals that CKm (45 kDa) is abundant in
fractions 26-34. A longer exposure (5 minutes, bottom blot) reveals
additional CKm immunoreactive bands with higher molecular weights. A
species with an apparent molecular weight of 88 kDa is observed in
fractions 26 and 28, and a protein with an apparent molecular weight of 130
kDa is observed in fractions 30 and 32. The same high molecular weight CKm
proteins were also observed after Blue Sepharose fractionation of young and
aged mouse quadriceps samples.