Introduction
Renal cell carcinoma (RCC) is the most common type of urinary system cancer, accounting for approximately 80% of all cases of renal malignancies [1]. In 2020, the global incidence of kidney cancer was estimated to be 431,288, with 179,368 deaths [2]. Clear cell renal cell carcinoma (ccRCC) is the most common subtype of RCC, accounting for approximately 85% of all RCC cases, and is the primary cause of mortality [3]. Advanced ccRCC is associated with a 5-year survival rate of only 12%, whereas a 5-year survival rate of up to 90% can be achieved if the disease is diagnosed at an earlier stage [4]. Unfortunately, nearly 20% of patients with ccRCC are typically not diagnosed until a late stage of disease progression [5]. Treatment for ccRCC is multimodal and includes surgery, chemotherapy, radiotherapy, targeted therapy, and immunotherapy [6]. Given the limited efficacy of radiotherapy and chemotherapy in treating ccRCC, surgery remains the primary approach; however, as many as 30% of patients experience postoperative disease recurrence within five years [7]. Hence, exploring novel prognostic models, distinguishing high-risk patients, and developing precision medicine are crucial to aiding the prognosis and treatment of ccRCC.
Disulfides represent an essential class of dynamic and redox-responsive covalent bonds and have been detected in various proteins [8]. Under oxidative stress, thiols in the cellular environment are converted to disulfides, thereby providing protection to cellular DNA against oxidative damage [9]. Dynamic thiol–disulfide homeostasis status plays critical roles in antioxidant/detoxification, apoptosis, signal transduction, and the regulation of enzymatic activity [10]. Disruption of the thiol–disulfide homeostasis status has long been considered a deleterious process in the body [11]. Recent studies involving SLC7A11high cells under glucose starvation have elucidated a unique form of disulfide-related cell death distinct from apoptosis and ferroptosis, namely disulfidptosis; it is characterized by aberrant accumulation of intracellular disulfides [12]. The substantial accumulation of disulfide molecules results in anomalous disulfide bonding among actin cytoskeleton proteins, which interferes with their tissues and ultimately culminates in the collapse of the actin network and cell death [12]. Disulfide dysregulation has been implicated in the pathogenesis of various diseases, including cancer [13]. However, the relationship between disulfidptosis and the pathogenesis and clinical outcomes associated with ccRCC remains unclear. Therefore, exploring the prognostic value and molecular mechanisms of disulfidptosis is crucial for the prognosis of ccRCC and the development of therapeutic strategies against ccRCC.
In the present study, RNA sequencing and clinical information of patients with ccRCC were obtained from public databases. Subsequently, patients with ccRCC were classified into three distinct disulfidptosis clusters based on the expression patterns of 10 disulfidptosis-related molecules and similarly classified into three gene clusters based on highly correlated genes identified in weighted gene co-expression network analysis (WGCNA). A scoring system was developed to predict overall survival (OS), the response to immunotherapy, and drug sensitivity in individual patients. Most importantly, the mRNA expression of disulfidptosis-related molecules in distinct cell subtypes was validated through RNA isolation, quantitative reverse transcriptase polymerase chain reaction (qRT-PCR), and single-cell RNA sequencing (scRNA-seq) data, respectively.
Materials and Methods
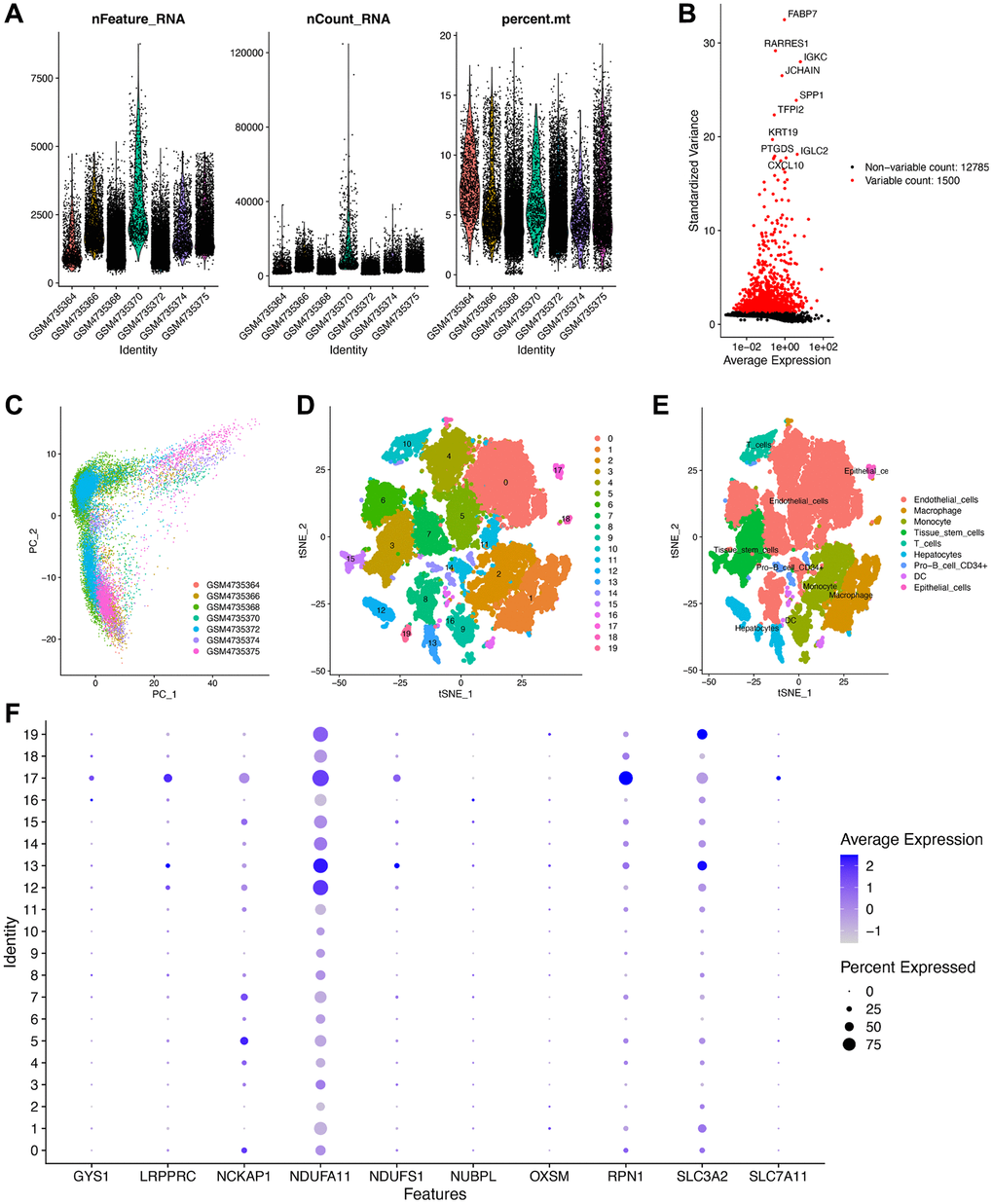
Data acquisition
RNA sequencing data (FPKM value) were acquired from The Cancer Genome Atlas (TCGA) and then converted into transcripts per million values. TCGA data pertaining to genome expression, variation, and correlation, were analyzed and visualized using the GSCALite website (http://bioinfo.life.hust.edu.cn/web/GSCALite/) [14]. The RNA sequencing data and clinical data of the RECA-EU (n = 91) and E-MTAB-1980 studies (n = 101) were downloaded from the International Cancer Genome Consortium (ICGC) and ArrayExpress database, respectively. Systematic batch effect correction was performed through the “ComBat” algorithm [15]. The median follow-up time was 38.75 months (range, 0–149.16 months) in this study. Additionally, the genomic and clinical data of two cohorts of patients who received immunotherapy, namely a cohort of patients with bladder cancer who received anti-programmed death ligand 1 (PD-L1) immunotherapy and a cohort of patients with metastatic melanoma who received anti-programmed death receptor-1 (PD-1) immunotherapy, were obtained from the IMvigor210 (http://research-pub.gene.com/IMvigor210CoreBiologies/) [16] and Gene Expression Omnibus (GEO, GSE78220, https://www.ncbi.nlm.nih.gov/gds/?term=GSE78220) databases, respectively [17]. Raw scRNA-seq data from seven ccRCC samples (GSE156632) sourced from the GEO database (https://www.ncbi.nlm.nih.gov/gds/?term=GSE156632) [18] was used to assess the expression levels of the disulfidptosis-related molecules in various cell subtypes. The aforementioned databases were publicly accessible and open-source. This study honors the data access policies of each database.
Identification of differentially expressed genes (DEGs) and WGCNA
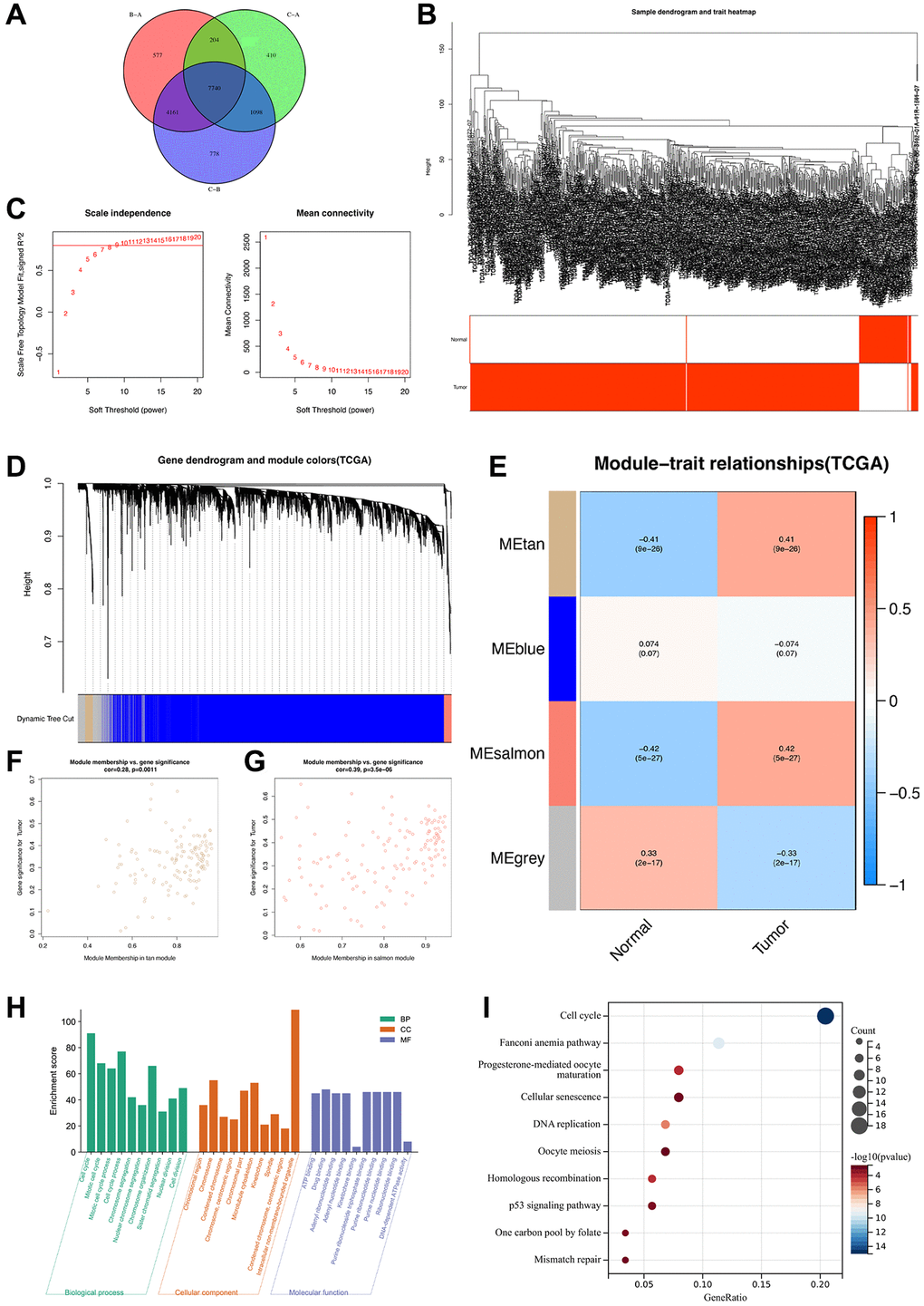
DEGs among the various clusters were identified using the “limma” package in R [23], with screening criteria of adjusted P < 0.001. The expression data of these DEGs were then employed to construct a gene co-expression network using the “weighted gene co-expression network analysis (WGCNA)” package in R [24]. Initially, both normal and ccRCC samples sourced from the TCGA database were analyzed, and the “hclust” function was used to remove outliers. Then, the optimal soft-thresholding power (β) was determined to allow scale-free topology with the “pickSoftThreshold” function. Thereafter, module eigengenes (MEs) were clustered to consolidate highly similar modules with a “mergeCutHeight” cutoff value of 0.25. To identify tumor-related modules, we calculated the correlation between the modules and clinical features (normal and tumor). The module with P < 0.05 was considered a gene module highly correlated with ccRCC, and the genes in the module were considered hub genes.
Functional enrichment analysis
To determine the potential functions and enriched pathways of the hub genes in the highly correlated gene modules, Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analyses were performed using the “Clusterprofiler” package in R [25]. P < 0.05 was considered to indicate statistically significant enrichment.
Construction of the disulfidptosis scoring system
To quantify the disulfidptosis status of individual patients, we developed a scoring system, namely the DSscore system, through principal component analysis (PCA). A univariate Cox analysis of OS was performed to identify the target genes with prognostic value (P < 0.05). We then used the target genes with prognostic value to perform PCA, and principal components 1 and 2 were designated as characteristic scores. A DSscore formula similar to that reported in previous studies was devised: DSscore = ∑(PC1i + PC2i). On the basis of the optimal cutoff value, we classified patients into low- and high-DSscore groups for subsequent analyses. Multivariate Cox regressions were applied to detect whether the DSscore was an independent prognostic indicator. Using the “rms” package in R [26], we then developed a nomogram to quantify the 1-, 3-, and 5-year survival outcomes in ccRCC. Additionally, to evaluate the reliability of the scoring system for various subgroups stratified by clinical parameters, we performed a stratified analysis.
Identification of immune status and tumor mutational burden (TMB) analysis
A correlation analysis was performed to illustrate the relationships between DSscores and tumor-infiltrating immune cells (TIICs). We then used the “maftools” package in R [27] to calculate TMB in ccRCC samples sourced from TCGA. The variance and correlation of TMB across the different DSscore groups were investigated through the Wilcoxon test and Spearman correlation analysis.
Immunotherapeutic efficacy and drug susceptibility analysis
The data of IMvigor210 and GSE78220 cohorts were used to evaluate the ability of DSscores to predict the response to immune checkpoint blockade treatment. Survival analyses were conducted using Kaplan-Meier survival curves and the log-rank test (P < 0.05). To determine the responses to chemotherapeutics, we determined the semi-inhibitory concentration (IC50) values of various drugs in the low- and high-DSscore groups using the “oncoPredict” package in R [28]. The differences in IC50 values between DSscore groups were examined with Wilcoxon signed-rank tests (P < 0.01).
Statistical analysis
All statistical analyses were performed using R, and P < 0.05 (two-tailed) was considered to indicate statistical significance in the absence of special annotation.
Data availability statement
The datasets analyzed in this study can be found in TCGA (http://www.cancer.gov/tcga), ICGC (https://dcc.icgc.org/projects/RECA-EU), ArrayExpress (https://www.ebi.ac.uk/biostudies/arrayexpress/), IMvigor210 (http://research-pub.gene.com/IMvigor210CoreBiologies/), and GEO (https://www.ncbi.nlm.nih.gov/gds/, GSE78220, and GSE156632) databases.
Results
Identification of gene clusters based on the hub genes
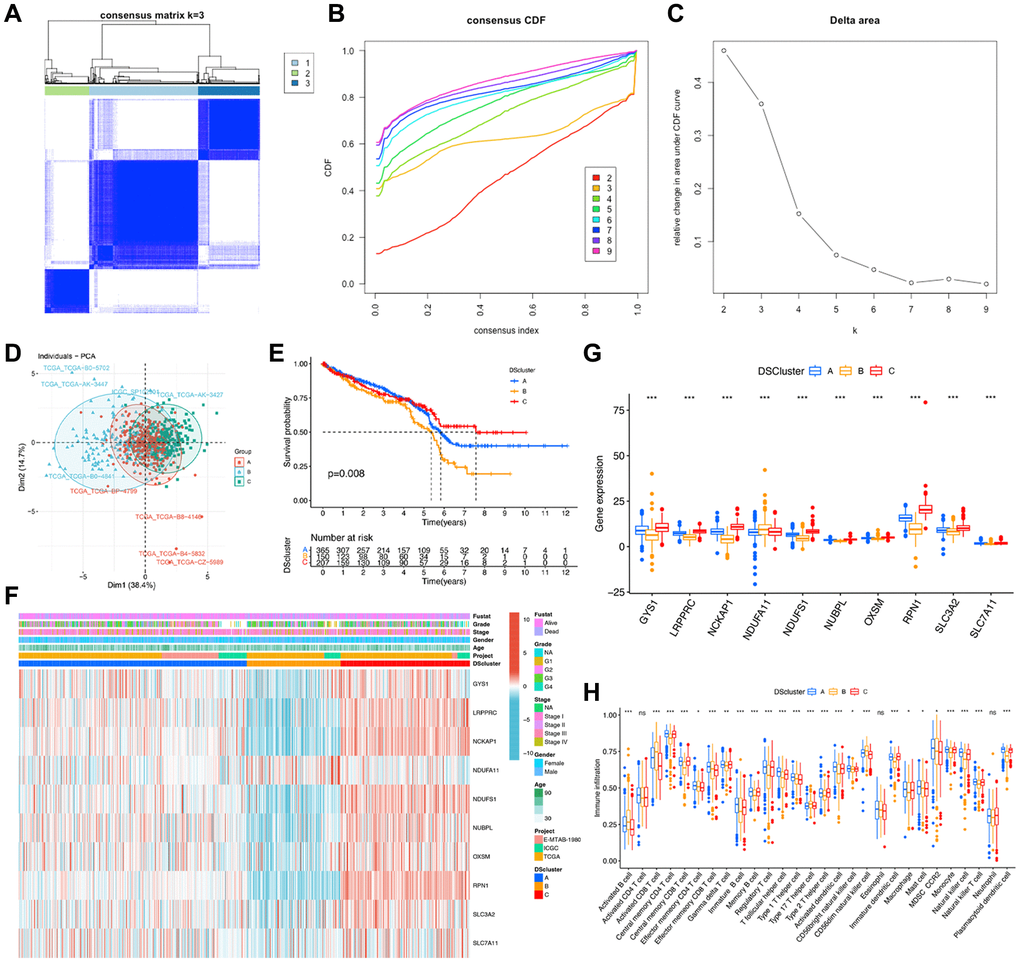
To further validate the regulation mechanism, we then identified different disulfidptosis-related gene clusters based on the expression of these 267 genes by WGCNA. The unsupervised clustering algorithm also classified patients with ccRCC into three distinct disulfidptosis-related gene clusters, namely geneCluster A, B, and C, consistent with the clustering based on disulfidptosis-related molecules (Figure 6A–6C) (Supplementary Table 2). This classification method was also validated by PCA (Figure 6D). Moreover, geneCluster C exhibited a particularly significant survival advantage, whereas geneCluster B exhibited the poorest prognosis (Figure 6E). A heat map revealed that most of the genes exhibited a gradual decrease in expression from geneCluster A to geneCluster C (Figure 6F). Seven disulfidptosis-related molecules were also significantly differentially expressed among the three geneClusters (Figure 6G), consistent with the results pertaining to the DSclusters.
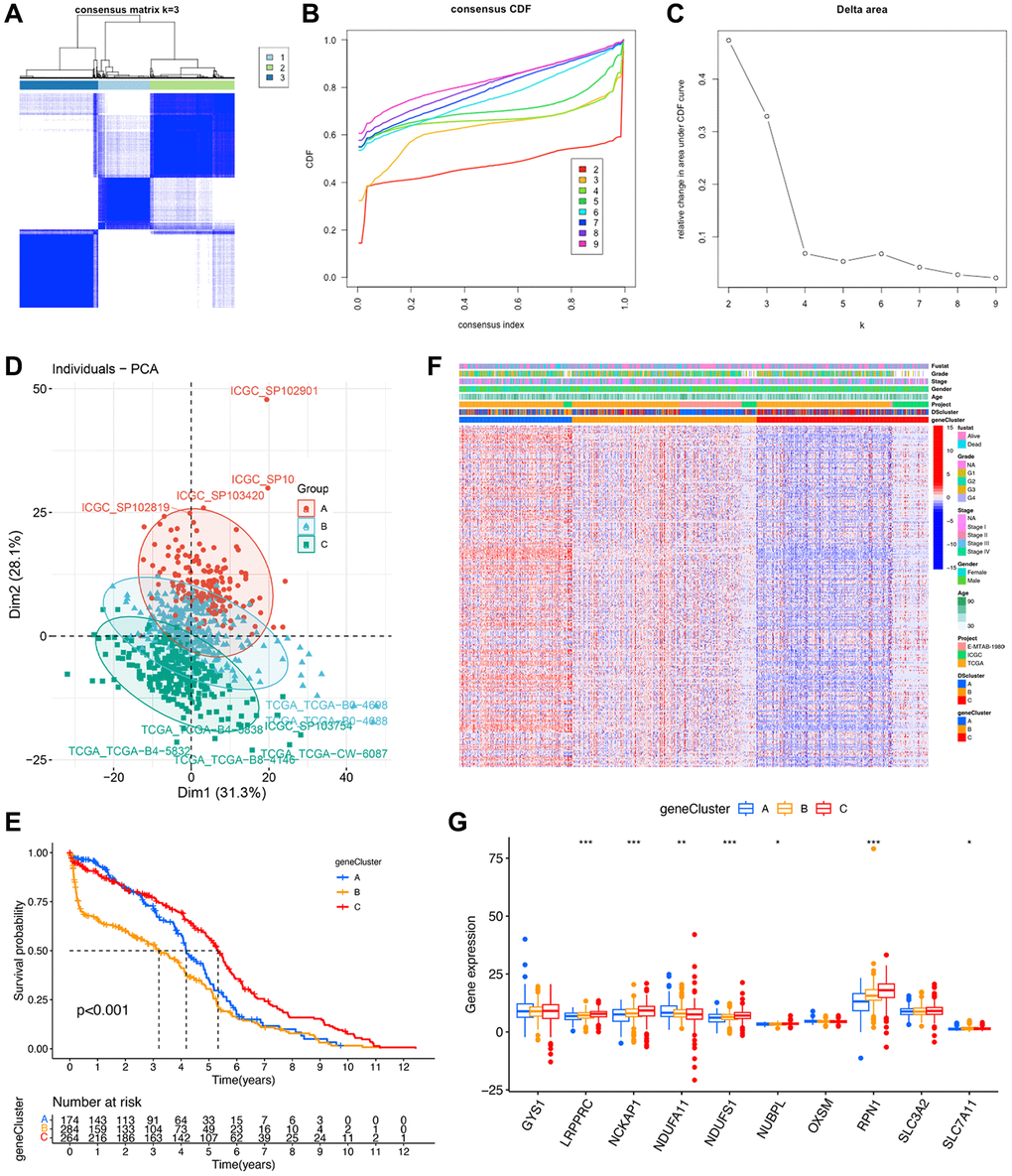
Figure 6. Identification of disulfidptosis gene clusters in ccRCC. (A) A color-coded heat map of the consensus matrix for k = 3. (B) Cumulative distribution function (CDF) curve of unsupervised clustering when k = 2–9. (C) Relative changes in area under the CDF curve when k = 2–9. (D) Principal component analysis showed a marked difference in the transcriptome between the three disulfidptosis gene clusters. (E) Kaplan–Meier survival curves of different disulfidptosis gene clusters. (F) Heat map of hub genes and clinicopathological features of patients in the three disulfidptosis gene clusters. (G) Expression of 10 disulfidptosis molecules in the three disulfidptosis gene clusters. *P < 0.05, **P < 0.01, ***P < 0.001.
Construction of a disulfidptosis scoring system
A total of 87 genes were identified as the potential prognostic genes in the tan and salmon modules in univariate Cox regression analysis. To further depict an individual’s disulfidptosis status, we established a risk score system, namely the DSscore system, via PCA of the 87 disulfidptosis cluster-related genes (Supplementary Table 2). As shown in the alluvial diagram that depicts the changes in the attributes of DScluster, geneCluster, DSscore, and survival status (Figure 7A), the DSscores were significantly different among the three different DSclusters. In detail, DScluster C exhibited the highest DSscore, followed by DSclusters A and B, in that order (Figure 7B). Significant differences in DSscores were also observed among the geneClusters (Figure 7C). The patients were then divided into two (low-DSscore and high-DSscore groups) according to a cutoff DSscore. The low-DSscore group exhibited considerably lower survival rates compared with the high-DSscore group (Figure 7D). Univariate and Multivariate analysis confirmed that stage, grade, and DSscore were an independent prognostic factor of OS in ccRCC (Figure 7E, 7F). A nomogram that included three independent clinical factors were then successfully established as a clinically applicable quantitative method for predicting individual OS (Figure 7G). The calibration curve confirmed the nomogram had optimal goodness of fit between the predicted and actual OS probabilities (Figure 7H). The ROC curve showed that the nomogram had a better discrimination ability (Figure 7I). Moreover, stratification analyses were performed to validate whether the DSscore retained its predictive ability in distinct subgroups. The results showed that compared with those with low DSscores, patients with high DSscores exhibited longer OS in all the subgroups, including age (age ≤60 and age >60), sex (female and male), stage (stage I and II and stage III and IV), and grade (grades 1–2 and grades 3–4) (Supplementary Figure 2). Collectively, these results indicated the feasibility of utilizing the DSscore-based classification method for predicting the OS of patients with ccRCC.
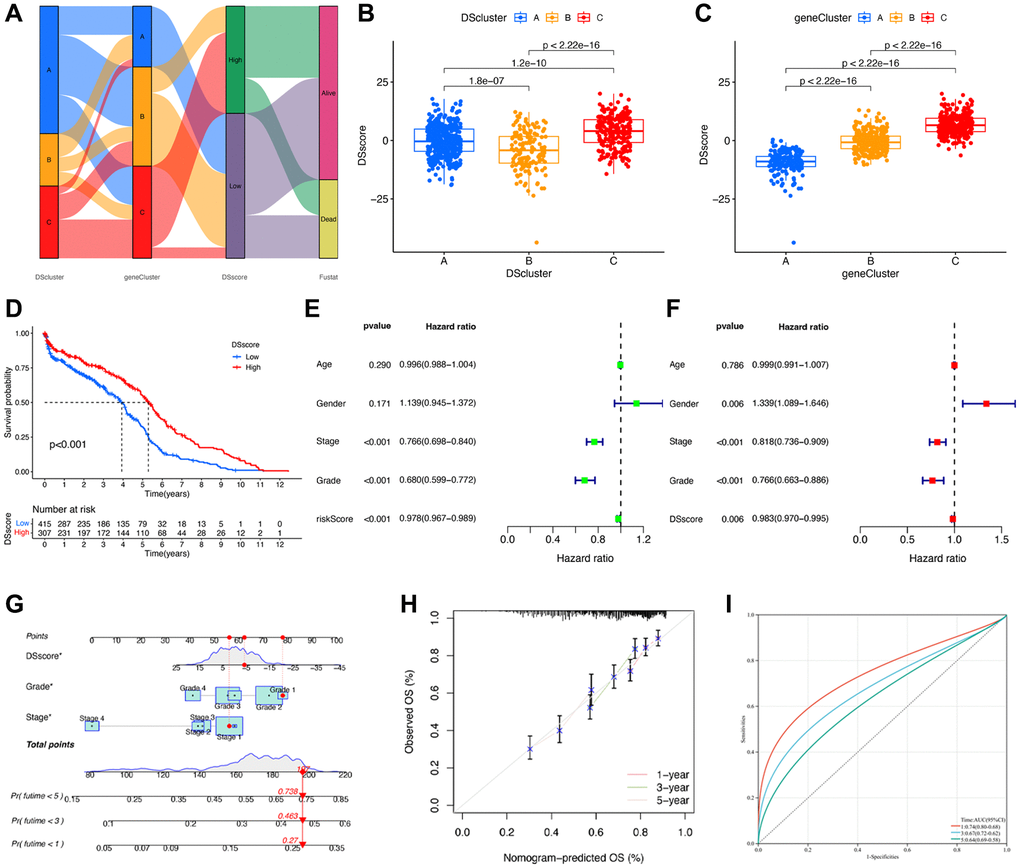
Figure 7. Construction of disulfidptosis score system. (A) Sankey plot of DScluster, geneCluster, DSscore, and survival status. (B) The differences in the DSscore among the three DSclusters. (C) The differences in the DSscore among the three disulfidptosis geneClusters. (D) Kaplan–Meier survival curves of low- and high-DSscore groups. (E) Univariate and (F) Multivariate Cox regression analyses of the DSscore and clinical features to determine prognostic value. (G) A comprehensive nomogram for predicting the survival probability of patients with ccRCC. (H) The calibration curve of the nomogram. (I) ROC curve of the nomogram. *P < 0.05.
DSscore is a predictor of the response to immunotherapy
We evaluated the correlation between DSscore and various immune cells and found a positive correlation between the DSscore and 21 of 28 immune-related cell infiltrates (Figure 8A), indicating a positive regulation of the immune reaction in patients with high DSscores. This finding was further emphasized by the difference in the immune cell profiles between the two DSscore groups (Figure 8B). Statically significant differences in the levels of TMB were observed between the two DSscore groups, and a positive correlation was observed between DSscores and TMB values. TMB analysis revealed that the TMB values were higher in the high-DSscore groups than those in the low-DSscore groups (Figure 8C). Further analyses revealed that TMB values were positively associated with DSscore ratings (Figure 8D). Survival analysis revealed that patients in the high-TMB group exhibited longer OS than that exhibited by patients in the low-TMB group (Figure 8E), suggesting that the prognosis of patients with ccRCC was influenced by TMB. Given that the inhibition of immunologic checkpoints has emerged as an unprecedented anticancer treatment, we then evaluated the prognostic value of DSscore in two cohorts who received immune checkpoint therapy (IMvigor210 cohort, anti-PD-L1 therapy; GSE78220 cohort; anti-PD-1 therapy). The patients in the IMvigor210 cohort and GSE78220 cohort were categorized into low- and high-DSscore groups. Patients in the high-DSscore group exhibited significantly longer OS compared with those in the low-DSscore group in both the IMvigor210 (P = 0.027, Figure 8F) and GSE78220 cohorts (P = 0.005, Figure 8G), suggesting the patients in the high-DSscore group exhibited superior immunotherapeutic repones. This finding was consistent with the finding that the high-TMB group was more likely to benefit from immunotherapy.
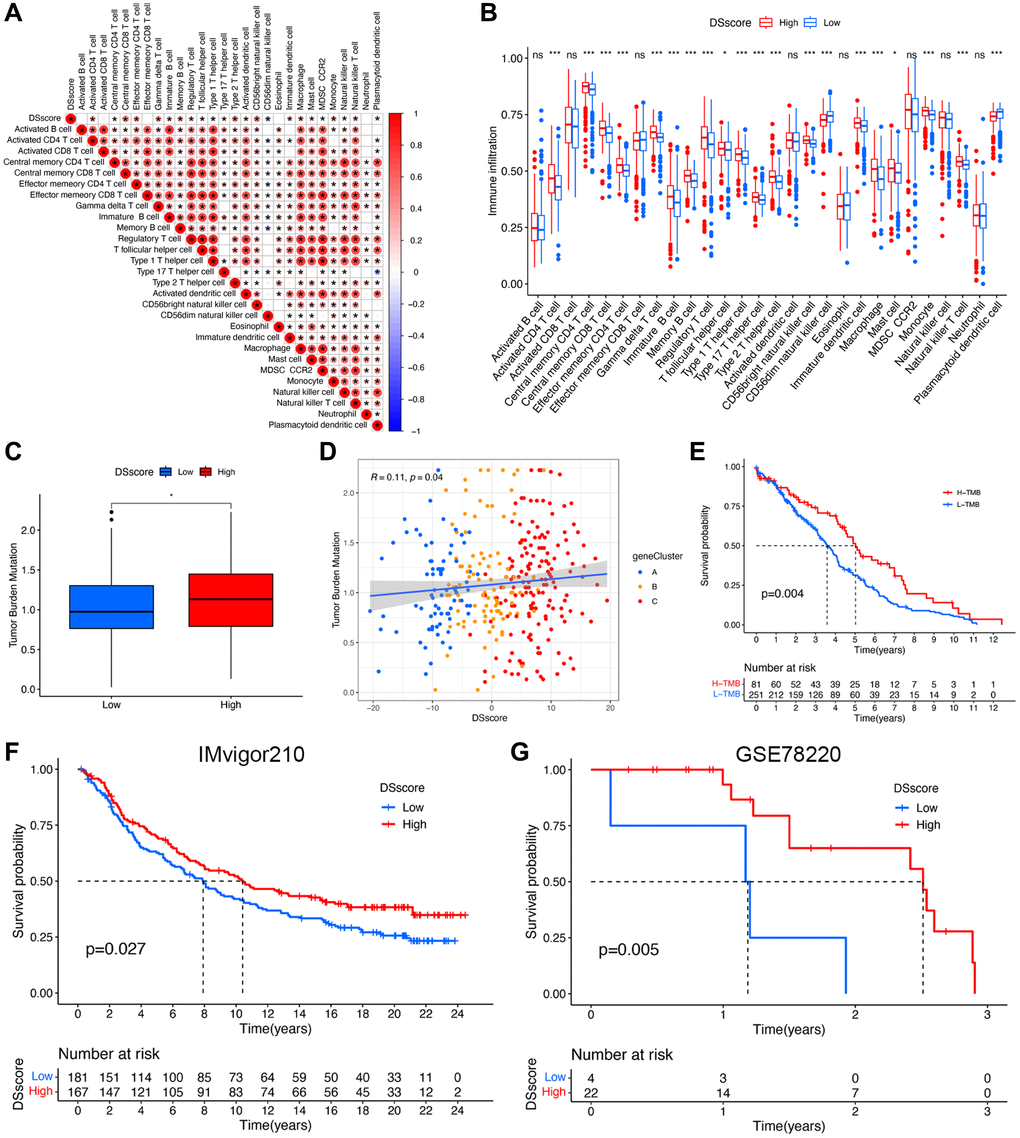
Figure 8. DSscore predicts the response to immunotherapy. (A) The correlation between DSscore and immune cell infiltration. (B) The differences in immune cell infiltration between the two DSscore groups. (C) The comparison of TMB between the two DSscore groups. (D) Correlation analysis of DSscore and TMB value. (E) Kaplan–Meier survival curves of the low- and high-TMB groups. (F) Kaplan–Meier survival curves of the low- and high-DSscore groups (IMvigor210 cohort). (G) Kaplan–Meier survival curves of low- and high-DSscore groups (GSE78220 cohort). *P < 0.05, ***P < 0.001. Abbreviation: ns: no significant.
DSscore is a predictor of drug sensitivity
To further examine the clinical utility of DSscore for precise ccRCC treatment, we evaluated the therapeutic efficacy of several commonly used chemotherapeutics in various DSscore groups. The results of analyses of IC50 values showed that patients with low DSscores were more responsive to MIRA-1, navitoclax, linsitinib, and PRIMA-1MET, whereas they were less sensitive to KU-55933 (Supplementary Figure 3).
Discussion
ccRCC is considered the most devastating tumor of the urinary system owing to its high incidence of recurrence and metastasis, as well as its insensitivity to chemoradiotherapy [31]. Previous studies have primarily focused on a single experimental study for ccRCC. However, few studies have reported satisfactory results. The extraction and identification of genetic data have garnered increasing attention in light of advancements in high-throughput sequencing and bioinformatics analysis. Programmed cell death (PCD) has been reported to play an essential role in ccRCC progression [32]. Many recent studies have developed prognostic signatures associated with PCD, such as cuproptosis [33], ferroptosis [34], and pyroptosis [35], for clinical guidance in ccRCC. Nevertheless, to date, the functions or signatures and the regulatory mechanisms of disulfidptosis in ccRCC remain unclear.
Disulfidptosis, a novel form of cell death reported in 2023 by Liu et al., has attracted the attention of researchers worldwide [12, 20]. A few research groups have elucidated that disulfidptosis plays an important role in various biological processes, such as homeostasis, may be involved in the development of tumors, and may potentially be used for the treatment of tumors [12, 36]. Disulfidptosis represents a mode of cystine accumulation in SLC7A11high cells subjected to glucose starvation and is distinct from other known types of PCDs. The anomalous disulfide bonds between actin cytoskeleton proteins contribute to the collapse of the actin network, which results in alterations in the localization and organization of glycolytic pathway members [12]. Owing to the unique metabolic characteristics of disulfidptosis, ccRCC is more closely associated with disulfidptosis compared to any other type of PCD [37]. In light of this, we conducted an integrative bioinformatics analysis to identify the potential prognostic value of disulfidptosis in ccRCC and disulfidptosis-related targets for ccRCC therapy.
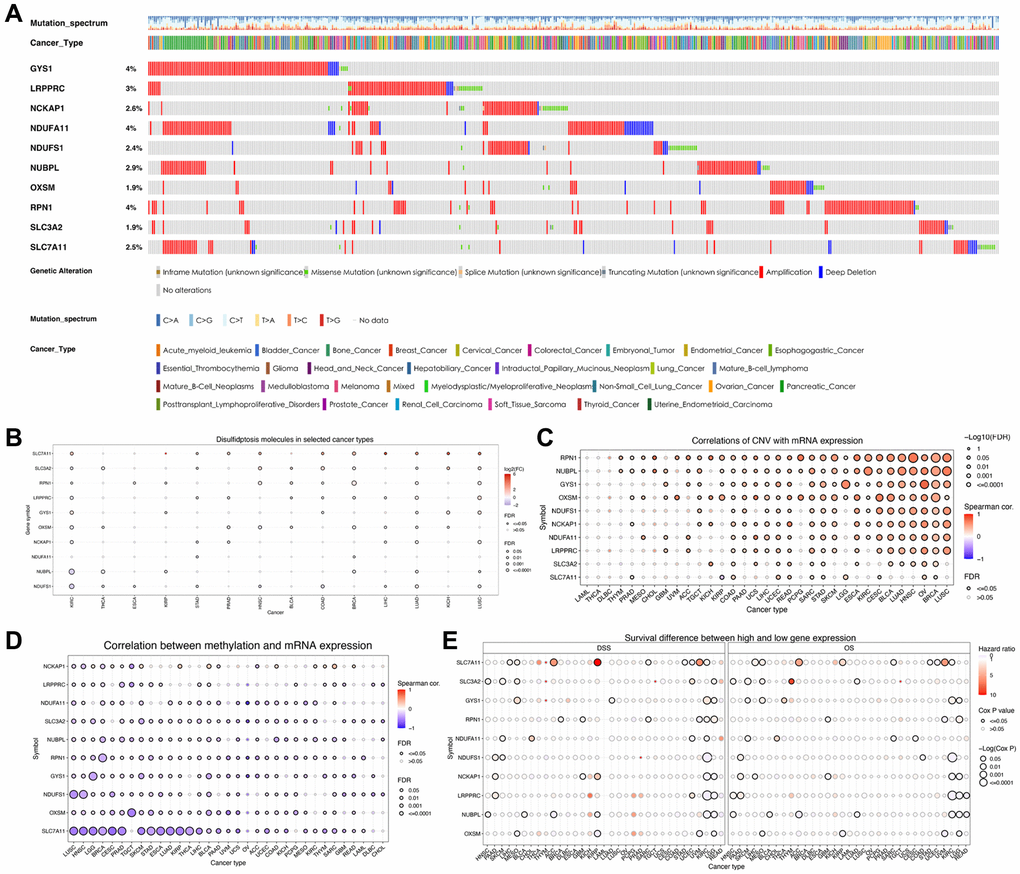
In the present study, we observed that most disulfidptosis-related molecules (9/10) were differentially expressed in ccRCC compared with normal tissues, suggesting a strong correlation between disulfidptosis and ccRCC progression. Based on the expression profiles of disulfidptosis-related molecules, the samples from the comprehensive cohort of TCGA–ccRCC, RECA-EU, and E-MTAB-1980 were classified into three clusters via unsupervised clustering. We then evaluated the levels of TIICs using the ssGSEA method and observed significant differences in the relative abundance of immune cells across the three subgroups. In particular, in DScluster B, the majority of the cells exhibited a notable decrease in immune cell infiltration compared with the other two DSclusters. Unexpectedly, however, DScluster B exhibited the highest count of active CD8 + T cells. In a study of melanoma, enhanced CD8 + T cell infiltration in TME exhibited a correlation with shorter OS [38]. In addition, the extent of CD8 + T cell infiltration in tumor tissues is consistent with the mutation and evolution of tumor cells, thereby contributing to tumor immune escape. To a certain extent, this also elucidates the inferior prognosis of patients in DScluster B. To analyze the downstream genes of the disulfidptosis, we conducted a differential analysis for the three DSclusters and identified 7,740 DEGs. WGCNA revealed 267 hub genes in the tan and salmon modules that were highly correlated with KIR. Enrichment analysis showed that hub genes were associated with the microtubule cytoskeleton and various signaling pathways, such as cell cycle, cellular senescence, and the p53 signaling pathway [12]. The close association of cell cycle and cellular senescence with cell death is well established [39, 40]. In ccRCC, the p53 pathway has been found to exhibit recurrent mutations [41]. We then determined two new gene clusters using the 267 hub genes. The results revealed that geneCluster B exhibited the worst prognosis. These findings provided more insights into the potential molecular mechanisms of disulfidptosis that may have therapeutic implications.
To quantify the status of disulfidptosis in individuals, we developed a scoring system, namely the DSscore system, using prognostic genes to define distinct disulfidptosis groups, thereby elucidating a means to predict the prognosis of ccRCC and guide clinical decision-making with increased accuracy. We calculated DSscores by PCA, and based on the optimal DSscore values, we classified patients with ccRCC into two groups, namely the low- and high-DSscore groups. The patients in the high-DSscore group exhibited particularly significant prolonged OS. Moreover, a significantly positive correlation was observed between the DSscores and most immune cells, consistent with the results of previous studies. For example, T cells constitute the predominant immune cell population within the TME, and their activity ratio is more strongly associated with the effective anti-tumor response in ccRCC [42]. Many studies have elucidated that decreased T cells might reflect the increased risk of ccRCC recurrence [43]. Research has elucidated a positive correlation exists between the proportion of NK cells in ccRCC and a favorable prognosis [44]. Moreover, a few patients with metastatic ccRCC have been observed to not respond to systemic cytokine treatment, which may be attributed, to a notable extent, to the lack of NK cell activation [45]. Generally, plasmacytoid dendritic cells are thought to promote cancer progression and tumor-induced tolerance [46]. The differences in outcomes between different DSscore groups are probably due to the distinct patterns of immune cell abundance. Thus, the potential mechanism of disulfidptosis affecting the progression of ccRCC may be associated with the regulation of immune cell infiltration.
Multiple clinical studies of solid tumors have elucidated that TMB levels may be a promising marker of immunotherapy response, with high TMB indicating better clinical response and longer OS [47–49]. For example, a higher level of TMB has been shown to be associated with a higher probability of a positive response to immune checkpoint inhibitors in patients with lung cancer via targeted next-generation sequencing [49]. In this study, patients in the high-DSscore group exhibited increased TMB, which is likely to be associated with longer survival outcomes. To validate our conjecture, we then explore the prognostic value of DSscore in two cohorts that received immune checkpoint therapy (IMvigor210 and GSE78220 cohorts). Following treatment, patients in the high-DSscore group exhibited significantly longer OS than those in the low-DSscore group, revealing that immunotherapy was one of the appropriate and clinically applicable anticancer strategies for patients with a high DSscore.
Drug susceptibility analysis showed that patients in the low-DSscore group were more sensitive to MIRA-1, navitoclax, linsitinib, and PRIMA-1MET compared with those in the high-DSscore group. The dysregulation of the p53 pathway results in the uncontrolled proliferation of tumor cells. Thus, the resumption of p53 functions, especially p53-dependent apoptosis, is a highly promising approach to anticancer treatment [50]. In the past years, several small molecules that target mut-p53-reactivation, including MIRA-1 and PRIMA-1MET, have been developed [51, 52]. Joerger and Fersht elucidated that MIRA-1 induces p53-independent apoptosis in multiple myeloma cells [51]. Recently, PRIMA-1MET was shown to limit the growth of colorectal cancer cells, irrespective of their p53 status [52]. Navitoclax, a member of the B-cell lymphoma 2 (BCL-2) family of protein inhibitors, was shown to induce apoptosis in cancer cells by disrupting the interactions between BCL-2-like proteins and BH3 domains [53]. The efficacy of navitoclax in the treatment of lung cancer and various relapsed or refractory lymphoid tumors has been elucidated by several phase I clinical studies [53, 54]. As an inhibitor of the insulin-like growth factor 1 receptor (IGF-1R), linsitinib has been identified as a satisfactory chemotherapeutic agent for multiple tumors [55]. Excitedly, in a recent study, the inhibition of IGF-1R using linsitinib reversed oncogenic function caused by the loss and/or downregulation of methylthioadenosine phosphorylase in RCC [56]. These drugs exerted an obvious inhibitory effect on tumor growth and are undoubtedly a promising therapeutic option for the patients in the low-DSscore group.
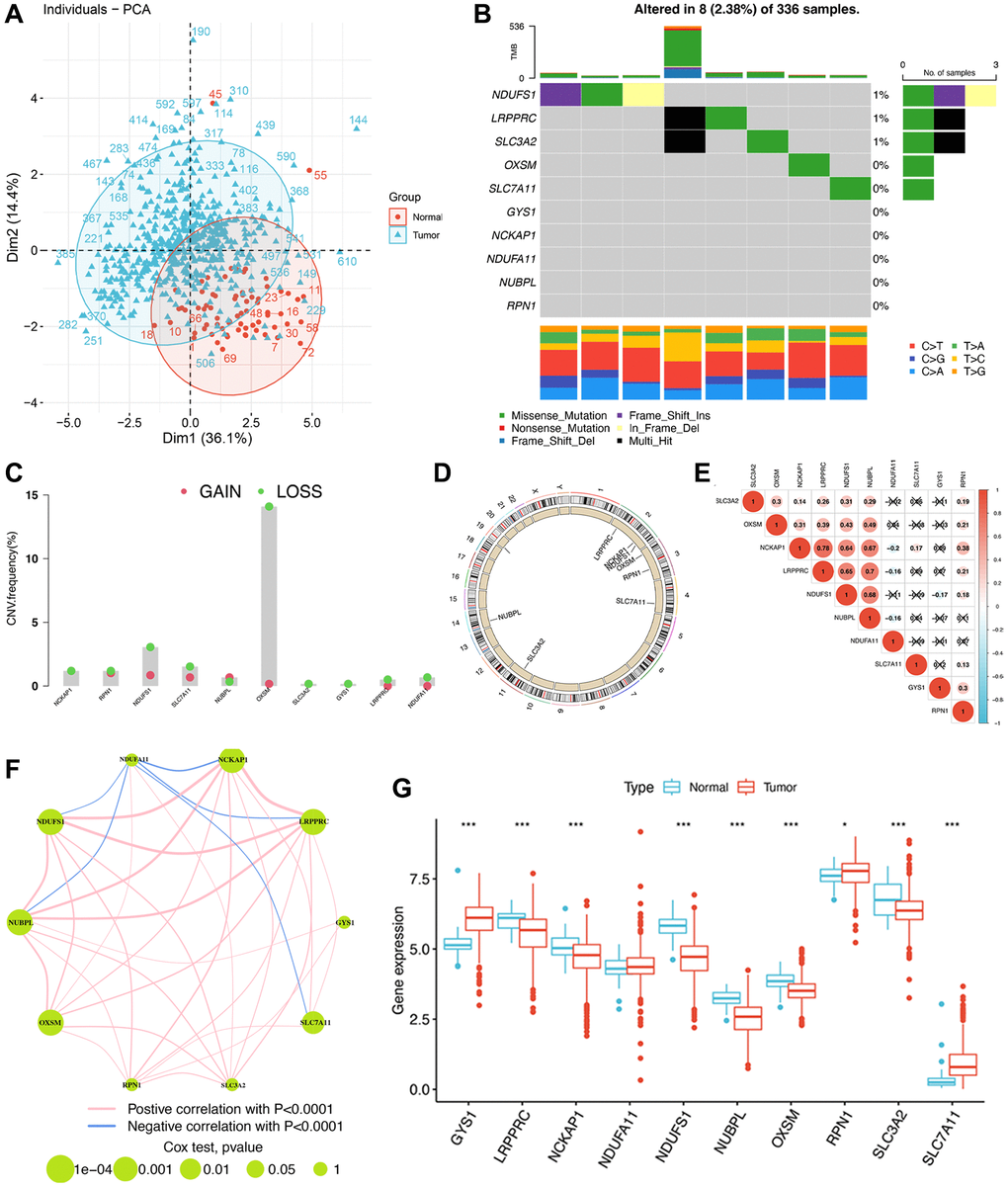
Finally, by performing qRT-PCR, we validated that the expression of GYS1 and SLC7A11 were upregulated, whereas those of NCKAP1, NDUFS1, NUBPL, OXSM, RPN1, and SLC3A2 were downregulated in ccRCC compared with those in the para-carcinoma tissues. Furthermore, we used scRNA-seq data from seven ccRCC samples to analyze cellular heterogeneity. The results also validated the possibility of disulfidptosis-related molecules in discriminating cell subtypes.
This study involved a comprehensive analysis of the association between disulfidptosis and ccRCC. However, several inevitable limitations existed. First, this study was conducted retrospectively using public databases, which may introduce a certain offset in the data. Second, we investigated the relationship between disulfidptosis and TME in ccRCC; however, we did not elucidate the underlying mechanisms by which disulfidptosis affects immune cells. Third, although qRT-PCR was performed to examine the expression of disulfidptosis-related molecules, additional in vitro or in vivo experiments are required to elucidate the mechanisms underlying disulfidptosis in ccRCC.
Conclusion
In summary, in this study, we elucidated three distinct disulfidptosis clusters in ccRCC and established a disulfidptosis scoring system for individuals. DSscore grouping has the potential to predict survival outcomes of patients and distinguish immune and clinicopathological characteristics. We also determined the therapeutic liability of DSscore in immunotherapy and chemotherapy; however, further studies are required to validate our findings. Overall, our findings provide a basis and elucidate the potential predictive value of the possible mechanisms underlying the correlation between disulfidptosis and ccRCC. Our findings may contribute to the development of novel clinical interventions.
Author Contributions
Weiming Deng designed the present study, interpreted the data, and performed the experimental validation. Zhenwei Xie and Libo Chen acquired and analyzed data. Wenjin Li drafted the manuscript. Wenjin Li and Mingyong Li revised the manuscript. All authors contributed to the article and approved the submitted version.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement and Consent
This study was approved by the Ethics and Human Subject Committee of the First Affiliated Hospital of the University of South China (IRB: [2022]02-126) and was conducted according to the guidelines of the Declaration of Helsinki. Written informed consent was obtained from all patients.
Funding
This work was funded by the Clinical Research Project of the University of South China (USCKF201902K01).
References
- 1. Hsieh JJ, Purdue MP, Signoretti S, Swanton C, Albiges L, Schmidinger M, Heng DY, Larkin J, Ficarra V. Renal cell carcinoma. Nat Rev Dis Primers. 2017; 3:17009. https://doi.org/10.1038/nrdp.2017.9 [PubMed]
- 2. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021; 71:209–49. https://doi.org/10.3322/caac.21660 [PubMed]
- 3. Bui TO, Dao VT, Nguyen VT, Feugeas JP, Pamoukdjian F, Bousquet G. Genomics of Clear-cell Renal Cell Carcinoma: A Systematic Review and Meta-analysis. Eur Urol. 2022; 81:349–61. https://doi.org/10.1016/j.eururo.2021.12.010 [PubMed]
- 4. Atkins MB, Tannir NM. Current and emerging therapies for first-line treatment of metastatic clear cell renal cell carcinoma. Cancer Treat Rev. 2018; 70:127–37. https://doi.org/10.1016/j.ctrv.2018.07.009 [PubMed]
- 5. Mitchell TJ, Turajlic S, Rowan A, Nicol D, Farmery JHR, O'Brien T, Martincorena I, Tarpey P, Angelopoulos N, Yates LR, Butler AP, Raine K, Stewart GD, et al, and TRACERx Renal Consortium. Timing the Landmark Events in the Evolution of Clear Cell Renal Cell Cancer: TRACERx Renal. Cell. 2018; 173:611–23.e17. https://doi.org/10.1016/j.cell.2018.02.020 [PubMed]
- 6. Thoma C. Kidney cancer: Combining targeted and immunotherapy. Nat Rev Urol. 2018; 15:263. https://doi.org/10.1038/nrurol.2018.43 [PubMed]
- 7. Dabestani S, Marconi L, Kuusk T, Bex A. Follow-up after curative treatment of localised renal cell carcinoma. World J Urol. 2018; 36:1953–9. https://doi.org/10.1007/s00345-018-2338-z [PubMed]
- 8. Brülisauer L, Gauthier MA, Leroux JC. Disulfide-containing parenteral delivery systems and their redox-biological fate. J Control Release. 2014; 195:147–54. https://doi.org/10.1016/j.jconrel.2014.06.012 [PubMed]
- 9. Karatas F, Acat M, Sahin S, Inci F, Karatas G, Neselioglu S, Haskul I, Erel O. The prognostic and predictive significance of serum thiols and disulfide levels in advanced non-small cell lung cancer. Aging Male. 2020; 23:619–28. https://doi.org/10.1080/13685538.2018.1559805 [PubMed]
- 10. Circu ML, Aw TY. Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic Biol Med. 2010; 48:749–62. https://doi.org/10.1016/j.freeradbiomed.2009.12.022 [PubMed]
- 11. Erel O, Neselioglu S. A novel and automated assay for thiol/disulphide homeostasis. Clin Biochem. 2014; 47:326–32. https://doi.org/10.1016/j.clinbiochem.2014.09.026 [PubMed]
- 12. Liu X, Nie L, Zhang Y, Yan Y, Wang C, Colic M, Olszewski K, Horbath A, Chen X, Lei G, Mao C, Wu S, Zhuang L, et al. Actin cytoskeleton vulnerability to disulfide stress mediates disulfidptosis. Nat Cell Biol. 2023; 25:404–14. https://doi.org/10.1038/s41556-023-01091-2 [PubMed]
- 13. Dirican N, Dirican A, Sen O, Aynali A, Atalay S, Bircan HA, Oztürk O, Erdogan S, Cakir M, Akkaya A. Thiol/disulfide homeostasis: A prognostic biomarker for patients with advanced non-small cell lung cancer? Redox Rep. 2016; 21:197–203. https://doi.org/10.1179/1351000215Y.0000000027 [PubMed]
- 14. Adib E, Nassar AH, Akl EW, Abou Alaiwi S, Nuzzo PV, Mouhieddine TH, Sonpavde G, Haddad RI, Mouw KW, Giannakis M, Hodi FS, Shukla SA, Gusev A, et al. CDKN2A Alterations and Response to Immunotherapy in Solid Tumors. Clin Cancer Res. 2021; 27:4025–35. https://doi.org/10.1158/1078-0432.CCR-21-0575 [PubMed]
- 15. Müller C, Schillert A, Röthemeier C, Trégouët DA, Proust C, Binder H, Pfeiffer N, Beutel M, Lackner KJ, Schnabel RB, Tiret L, Wild PS, Blankenberg S, et al. Removing Batch Effects from Longitudinal Gene Expression - Quantile Normalization Plus ComBat as Best Approach for Microarray Transcriptome Data. PLoS One. 2016; 11:e0156594. https://doi.org/10.1371/journal.pone.0156594 [PubMed]
- 16. Mariathasan S, Turley SJ, Nickles D, Castiglioni A, Yuen K, Wang Y, Kadel EE
III , Koeppen H, Astarita JL, Cubas R, Jhunjhunwala S, Banchereau R, Yang Y, et al. TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature. 2018; 554:544–8. https://doi.org/10.1038/nature25501 [PubMed] - 17. Hugo W, Zaretsky JM, Sun L, Song C, Moreno BH, Hu-Lieskovan S, Berent-Maoz B, Pang J, Chmielowski B, Cherry G, Seja E, Lomeli S, Kong X, et al. Genomic and Transcriptomic Features of Response to Anti-PD-1 Therapy in Metastatic Melanoma. Cell. 2016; 165:35–44. https://doi.org/10.1016/j.cell.2016.02.065 [PubMed]
- 18. Zhang M, Zhai W, Miao J, Cheng X, Luo W, Song W, Wang J, Gao WQ. Single cell analysis reveals intra-tumour heterogeneity, microenvironment and potential diagnosis markers for clear cell renal cell carcinoma. Clin Transl Med. 2022; 12:e713. https://doi.org/10.1002/ctm2.713 [PubMed]
- 19. Zheng T, Liu Q, Xing F, Zeng C, Wang W. Disulfidptosis: a new form of programmed cell death. J Exp Clin Cancer Res. 2023; 42:137. https://doi.org/10.1186/s13046-023-02712-2 [PubMed]
- 20. Machesky LM. Deadly actin collapse by disulfidptosis. Nat Cell Biol. 2023; 25:375–6. https://doi.org/10.1038/s41556-023-01100-4 [PubMed]
- 21. Wilkerson MD, Hayes DN. ConsensusClusterPlus: a class discovery tool with confidence assessments and item tracking. Bioinformatics. 2010; 26:1572–3. https://doi.org/10.1093/bioinformatics/btq170 [PubMed]
- 22. Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub TR, Lander ES, Mesirov JP. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A. 2005; 102:15545–50. https://doi.org/10.1073/pnas.0506580102 [PubMed]
- 23. Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015; 43:e47. https://doi.org/10.1093/nar/gkv007 [PubMed]
- 24. Langfelder P, Horvath S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics. 2008; 9:559. https://doi.org/10.1186/1471-2105-9-559 [PubMed]
- 25. Yu G, Wang LG, Han Y, He QY. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS. 2012; 16:284–7. https://doi.org/10.1089/omi.2011.0118 [PubMed]
- 26. Liu Y, Li L, Jiang D, Yang M, Gao X, Lv K, Xu W, Wei H, Wan W, Xiao J. A Novel Nomogram for Survival Prediction of Patients with Spinal Metastasis From Prostate Cancer. Spine (Phila Pa 1976). 2021; 46:E364–73. https://doi.org/10.1097/BRS.0000000000003888 [PubMed]
- 27. Mayakonda A, Lin DC, Assenov Y, Plass C, Koeffler HP. Maftools: efficient and comprehensive analysis of somatic variants in cancer. Genome Res. 2018; 28:1747–56. https://doi.org/10.1101/gr.239244.118 [PubMed]
- 28. Maeser D, Gruener RF, Huang RS. oncoPredict: an R package for predicting in vivo or cancer patient drug response and biomarkers from cell line screening data. Brief Bioinform. 2021; 22:bbab260. https://doi.org/10.1093/bib/bbab260 [PubMed]
- 29. Macosko EZ, Basu A, Satija R, Nemesh J, Shekhar K, Goldman M, Tirosh I, Bialas AR, Kamitaki N, Martersteck EM, Trombetta JJ, Weitz DA, Sanes JR, et al. Highly Parallel Genome-wide Expression Profiling of Individual Cells Using Nanoliter Droplets. Cell. 2015; 161:1202–14. https://doi.org/10.1016/j.cell.2015.05.002 [PubMed]
- 30. Aran D, Looney AP, Liu L, Wu E, Fong V, Hsu A, Chak S, Naikawadi RP, Wolters PJ, Abate AR, Butte AJ, Bhattacharya M. Reference-based analysis of lung single-cell sequencing reveals a transitional profibrotic macrophage. Nat Immunol. 2019; 20:163–72. https://doi.org/10.1038/s41590-018-0276-y [PubMed]
- 31. Rini BI, Campbell SC, Escudier B. Renal cell carcinoma. Lancet. 2009; 373:1119–32. https://doi.org/10.1016/S0140-6736(09)60229-4 [PubMed]
- 32. Bedoui S, Herold MJ, Strasser A. Emerging connectivity of programmed cell death pathways and its physiological implications. Nat Rev Mol Cell Biol. 2020; 21:678–95. https://doi.org/10.1038/s41580-020-0270-8 [PubMed]
- 33. Zhang G, Chen X, Fang J, Tai P, Chen A, Cao K. Cuproptosis status affects treatment options about immunotherapy and targeted therapy for patients with kidney renal clear cell carcinoma. Front Immunol. 2022; 13:954440. https://doi.org/10.3389/fimmu.2022.954440 [PubMed]
- 34. Sun Z, Li T, Xiao C, Zou S, Zhang M, Zhang Q, Wang Z, Zhan H, Wang H. Prediction of overall survival based upon a new ferroptosis-related gene signature in patients with clear cell renal cell carcinoma. World J Surg Oncol. 2022; 20:120. https://doi.org/10.1186/s12957-022-02555-9 [PubMed]
- 35. Khan M, Ai M, Du K, Song J, Wang B, Lin J, Ren A, Chen C, Huang Z, Qiu W, Zhang J, Tian Y, Yuan Y. Pyroptosis relates to tumor microenvironment remodeling and prognosis: A pan-cancer perspective. Front Immunol. 2022; 13:1062225. https://doi.org/10.3389/fimmu.2022.1062225 [PubMed]
- 36. Sezgin B, Kinci MF, Pirinççi F, Camuzcuoğlu A, Erel Ö, Neşelioğlu S, Camuzcuoğlu H. Thiol-disulfide status of patients with cervical cancer. J Obstet Gynaecol Res. 2020; 46:2423–29. https://doi.org/10.1111/jog.14480 [PubMed]
- 37. Linehan WM, Srinivasan R, Schmidt LS. The genetic basis of kidney cancer: a metabolic disease. Nat Rev Urol. 2010; 7:277–85. https://doi.org/10.1038/nrurol.2010.47 [PubMed]
- 38. Tumeh PC, Harview CL, Yearley JH, Shintaku IP, Taylor EJ, Robert L, Chmielowski B, Spasic M, Henry G, Ciobanu V, West AN, Carmona M, Kivork C, et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature. 2014; 515:568–71. https://doi.org/10.1038/nature13954 [PubMed]
- 39. Wiman KG, Zhivotovsky B. Understanding cell cycle and cell death regulation provides novel weapons against human diseases. J Intern Med. 2017; 281:483–95. https://doi.org/10.1111/joim.12609 [PubMed]
- 40. Shen S, Ji C, Wei K. Cellular Senescence and Regulated Cell Death of Tubular Epithelial Cells in Diabetic Kidney Disease. Front Endocrinol (Lausanne). 2022; 13:924299. https://doi.org/10.3389/fendo.2022.924299 [PubMed]
- 41. Sato Y, Yoshizato T, Shiraishi Y, Maekawa S, Okuno Y, Kamura T, Shimamura T, Sato-Otsubo A, Nagae G, Suzuki H, Nagata Y, Yoshida K, Kon A, et al. Integrated molecular analysis of clear-cell renal cell carcinoma. Nat Genet. 2013; 45:860–7. https://doi.org/10.1038/ng.2699 [PubMed]
- 42. Giraldo NA, Becht E, Vano Y, Petitprez F, Lacroix L, Validire P, Sanchez-Salas R, Ingels A, Oudard S, Moatti A, Buttard B, Bourass S, Germain C, et al. Tumor-Infiltrating and Peripheral Blood T-cell Immunophenotypes Predict Early Relapse in Localized Clear Cell Renal Cell Carcinoma. Clin Cancer Res. 2017; 23:4416–28. https://doi.org/10.1158/1078-0432.CCR-16-2848 [PubMed]
- 43. Ghatalia P, Gordetsky J, Kuo F, Dulaimi E, Cai KQ, Devarajan K, Bae S, Naik G, Chan TA, Uzzo R, Hakimi AA, Sonpavde G, Plimack E. Prognostic impact of immune gene expression signature and tumor infiltrating immune cells in localized clear cell renal cell carcinoma. J Immunother Cancer. 2019; 7:139. https://doi.org/10.1186/s40425-019-0621-1 [PubMed]
- 44. Eckl J, Buchner A, Prinz PU, Riesenberg R, Siegert SI, Kammerer R, Nelson PJ, Noessner E. Transcript signature predicts tissue NK cell content and defines renal cell carcinoma subgroups independent of TNM staging. J Mol Med (Berl). 2012; 90:55–66. https://doi.org/10.1007/s00109-011-0806-7 [PubMed]
- 45. Toliou T, Stravoravdi P, Polyzonis M, Vakalikos J. Natural killer cell activation after interferon administration in patients with metastatic renal cell carcinoma: an ultrastructural and immunohistochemical study. Eur Urol. 1996; 29:252–6. [PubMed]
- 46. Verneau J, Sautés-Fridman C, Sun CM. Dendritic cells in the tumor microenvironment: prognostic and theranostic impact. Semin Immunol. 2020; 48:101410. https://doi.org/10.1016/j.smim.2020.101410 [PubMed]
- 47. Samstein RM, Lee CH, Shoushtari AN, Hellmann MD, Shen R, Janjigian YY, Barron DA, Zehir A, Jordan EJ, Omuro A, Kaley TJ, Kendall SM, Motzer RJ, et al. Tumor mutational load predicts survival after immunotherapy across multiple cancer types. Nat Genet. 2019; 51:202–6. https://doi.org/10.1038/s41588-018-0312-8 [PubMed]
- 48. Liu L, Bai X, Wang J, Tang XR, Wu DH, Du SS, Du XJ, Zhang YW, Zhu HB, Fang Y, Guo ZQ, Zeng Q, Guo XJ, et al. Combination of TMB and CNA Stratifies Prognostic and Predictive Responses to Immunotherapy Across Metastatic Cancer. Clin Cancer Res. 2019; 25:7413–23. https://doi.org/10.1158/1078-0432.CCR-19-0558 [PubMed]
- 49. Rizvi H, Sanchez-Vega F, La K, Chatila W, Jonsson P, Halpenny D, Plodkowski A, Long N, Sauter JL, Rekhtman N, Hollmann T, Schalper KA, Gainor JF, et al. Molecular Determinants of Response to Anti-Programmed Cell Death (PD)-1 and Anti-Programmed Death-Ligand 1 (PD-L1) Blockade in Patients With Non-Small-Cell Lung Cancer Profiled With Targeted Next-Generation Sequencing. J Clin Oncol. 2018; 36:633–41. https://doi.org/10.1200/JCO.2017.75.3384 [PubMed]
- 50. Duffy MJ, Synnott NC, Crown J. Mutant p53 as a target for cancer treatment. Eur J Cancer. 2017; 83:258–65. https://doi.org/10.1016/j.ejca.2017.06.023 [PubMed]
- 51. Joerger AC, Fersht AR. Structural biology of the tumor suppressor p53. Annu Rev Biochem. 2008; 77:557–82. https://doi.org/10.1146/annurev.biochem.77.060806.091238 [PubMed]
- 52. Yu Y, Kalinowski DS, Kovacevic Z, Siafakas AR, Jansson PJ, Stefani C, Lovejoy DB, Sharpe PC, Bernhardt PV, Richardson DR. Thiosemicarbazones from the old to new: iron chelators that are more than just ribonucleotide reductase inhibitors. J Med Chem. 2009; 52:5271–94. https://doi.org/10.1021/jm900552r [PubMed]
- 53. Han Z, Liang J, Li Y, He J. Drugs and Clinical Approaches Targeting the Antiapoptotic Protein: A Review. Biomed Res Int. 2019; 2019:1212369. https://doi.org/10.1155/2019/1212369 [PubMed]
- 54. Wilson WH, O'Connor OA, Czuczman MS, LaCasce AS, Gerecitano JF, Leonard JP, Tulpule A, Dunleavy K, Xiong H, Chiu YL, Cui Y, Busman T, Elmore SW, et al. Navitoclax, a targeted high-affinity inhibitor of BCL-2, in lymphoid malignancies: a phase 1 dose-escalation study of safety, pharmacokinetics, pharmacodynamics, and antitumour activity. Lancet Oncol. 2010; 11:1149–59. https://doi.org/10.1016/S1470-2045(10)70261-8 [PubMed]
- 55. Guo Y, Mehrabi Nasab E, Hassanpour F, Athari SS. Linsitinib and aspirin as the IGF1-R antagonists, inhibit regorafenib-resistant chemotherapy in colon cancer. Saudi J Biol Sci. 2022; 29:872–7. https://doi.org/10.1016/j.sjbs.2021.10.019 [PubMed]
- 56. Xu J, Chang WH, Fong LWR, Weiss RH, Yu SL, Chen CH. Targeting the insulin-like growth factor-1 receptor in MTAP-deficient renal cell carcinoma. Signal Transduct Target Ther. 2019; 4:2. https://doi.org/10.1038/s41392-019-0035-z [PubMed]